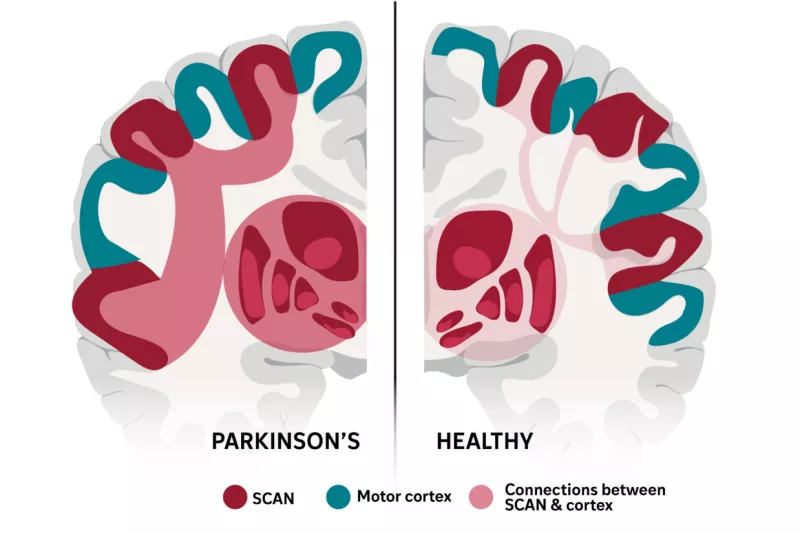
FDA Rejects Moderna mRNA Flu Vaccine Filing
The U.S. drug regulator has declined to review Moderna’s application for its first seasonal influenza vaccine based on mRNA technology, marking a significant hurdle for a platform once celebrated for its rapid pandemic response. The decision, disclosed by the company, centers on questions about the structure of its clinical trial